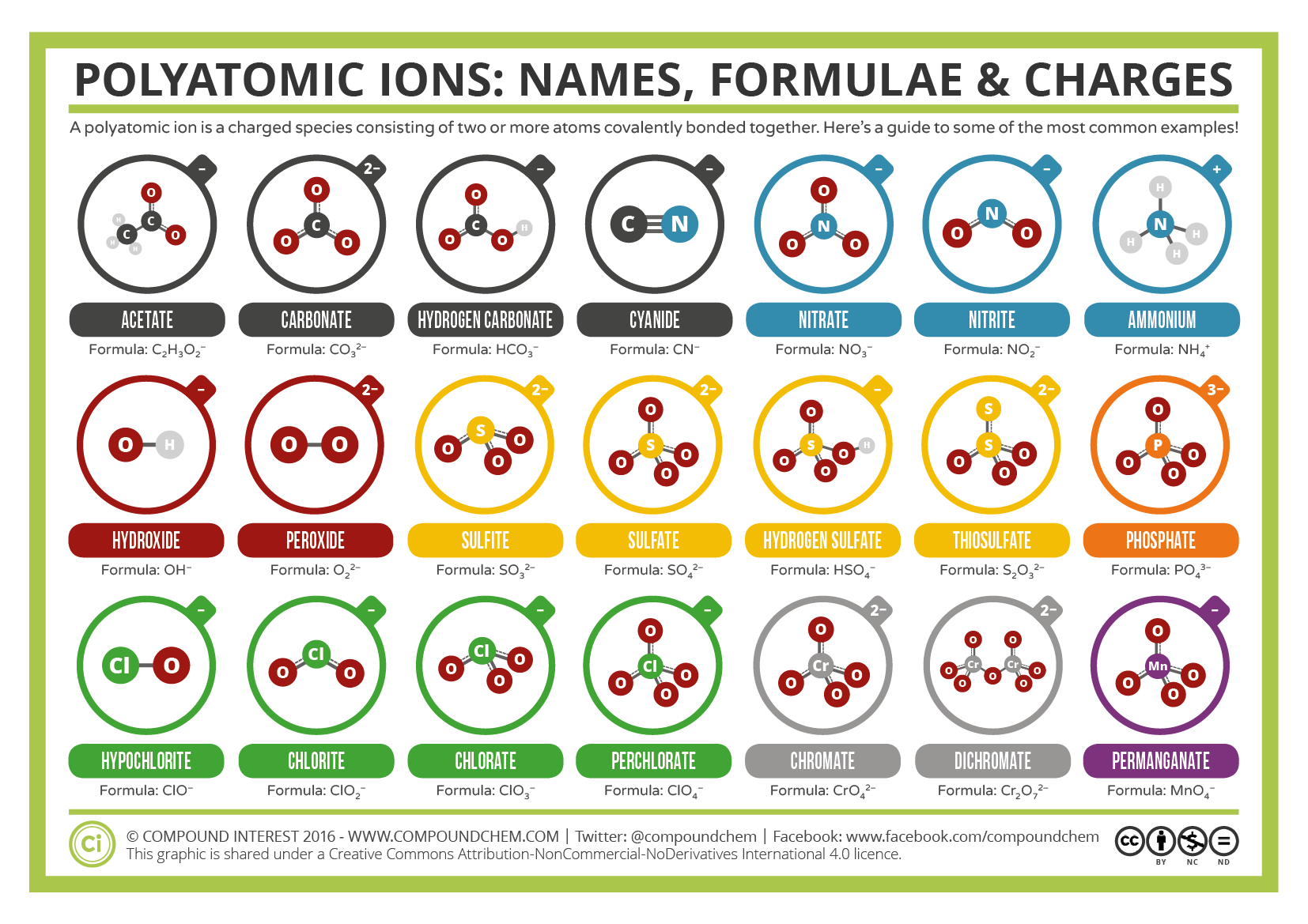
Similarly, we can think of a polyatomic ion as a molecule that has been ionized by gaining or losing electrons. charges of the most common polyatomic ions. Since there is one extra electron compared to the number of protons, the ion has a net charge of 1. Such acids include sulfuric acid (H 2 SO 4 ) or carbonic acid (H 2 CO 3 ). Note the usefulness of the periodic table in predicting likely ion formation and charge ((Figure)). Table: Common Polyatomic ion the topic of acids and polyatomic ions, there is nomenclature of aqueous acids. 
To correctly specify how many oxygen atoms are in the ion, prefixes and suffixes are again used. periodic table to predict the sign and the charge on the lithium ion.). This occurs because the number of oxygen atoms are increasing from hypochlorite to perchlorate, yet the overall charge of the polyatomic ion is still -1. charges of the most common polyatomic ions (Table 1). (Usage of this example can be seen from the set of compounds containing Cl and O) Increasing oxidation state of the nonmetal Hypo chlorite chlorite chlorate per chlorateĪs indicated by the arrow, moving to the right, the following trends occur: So, this makes the ion have a negative charge.\): Slide showing periodic trends for nomenclature of oxyanions of halogens.įigure 2.7.4 Shows several types of polyatomic anions, name When we gain more electrons, we can know that the amount of electrons is higher than the proton. Whether an atom forms a cation or an anion depends on its position. But the valency of elements, when combined with H or O first, increases from 1 to 4 and then it reduces to zero. While moving left to right across a period, the number of valence electrons of elements increases and varies between 1 to 8. Variation Of Oxidation State Along a Period. So, we can conclude that this ion has a positive charge and vice versa. Predicting Cations and Anions based on the Periodic Table. Periodic Trends in the Oxidation States of Elements. So, the dominant charge of the certain ion is the proton. Hopefully this helped, you can always add word to the sentence to help remember other polyatomic ions. Camel Carbonate 3 consonants 3 O's 2 vowels 2- COX3X2 C O X 3 X 2. If the ion loses the electron, this means that the negative charge is lesser than the positive charge (proton). The number of vowels stand for the charge. Actually, to understand the ion, we can see how the atoms lose their negative charge (electron). Actually, we have discussed this before above. When we download a periodic table of ions, we also need to know about how to determine an ion. The anion has the -ide ending for a binary compound or else a polyatomic ion name. The cation is the element name followed by a Roman numeral in parentheses if the element has multiple charges. When naming ionic compounds, list the cation first and the anion second. Then, if it is reversed, which means the amount of electrons is higher than the proton, we can call it anion. Naming Ionic Compounds Nomenclature Rules. The cation is where the positive charge (proton) has more amount than the electron. There are two kinds of ions in the periodic table of ions. However, it is pretty normal to be found that there is an imbalance of the charge, whether the number of protons is higher or the number of electrons is higher. Well, there are two kinds of ions on the periodic table.Īctually, if an atom or atoms that have equal numbers of negative charge (electrons) and positive charge (proton), then they can be defined as a neutral atom. Yes, one of the things that is required to be understood in the beginning is the ions themselves. The ions have the same magnitude of charge, one of each (ion) is needed to balance the charges. Carbonate (CO3) is the polyatomic ion and possesses a charge of -2 By crisscrossing these charges, the ratio of 2:2 reduces to a 1:1 ratio and the resulting formula would have no additional. Two ammonium ions need to balance the charge on a single sulfide ion. Note that only two polyatomic ions in this table are cations, hydronium ion (H 3 O +) and ammonium ion (NH 4+ ), the remaining. Table of Polyatomic Ions There are a number of ions that are not individual atoms but are composed of multiple atoms that are covalently bonded together. For example, NO3 NO 3 is the nitrate ion it has one nitrogen atom and three oxygen atoms and an overall 1 charge. Yes, we can call this a periodic table of ions.īefore we download the periodic table of irons, we actually need to understand the basic things inside the periodic table of ions. The ammonium ion has a 1+ charge and the sulfide ion has a 2 charge. 1 lists the ion names and ion formulas of the most common polyatomic ions. When it comes to learning Chemistry lessons, we may have seen our teacher introduce us with a sheet of the grid that consists of many abbreviations as a symbol of the elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
